Authenticity noble metals is usually certified by breakdownThe state authorities are responsible for this. At the same time, there are cases where it is necessary to check outwhether or not the gold object is a fake.
When is it necessary to check the authenticity of gold
There are many situations where you have to at home to establish whether we see natural gold or counterfeit metal in front of our eyes. Counterfeits of the gold often look like natural metal and represent alloy of several elements. Brass, aluminum, and iron are used for this purpose. Alloys are used: vermei, pinchbeck, batbronze. Sometimes, it is just well polished copper. Another option is when it can be said that precious metal is not authentic - the use of gold of a lower standard than the declared one.
Along with professional methods of verification using special chemical reagents and assay stones, as well as modern analyzers, there are many folk options.
The craftsmen come up with more and more new methods of testing the noble material for authenticity. There are known variants of identification with the use of vinegar, magnita, lapis pencil. One of the most reliable, reliable methods: check for gold Iodine at home. The explanation is that iodine is a rare chemical element that can react with such an inert element as zooloto.

Necessary tools for checking with iodine
In goes a simple technology using tools that can be found in any home. To test it, you'll need:
- alcoholic iodine solution, it is sold in different concentrations. The most common version is 5% iodine solution.
- cotton swab;
- sandpaper;
- high concentration ethyl alcohol;
- A napkin made of a soft cloth, such as flannel.
A magnifying glass and tweezers are good for the job if you need to check small parts.
Iodine is able to react with gold under normal conditions, it darkens at the application site. Gold iodide, AuI, is formed. The dark area will have different shades. It depends on the gold standard: 375, 585 or 750. The latter gives, having combined with iodide, a zone of dark green color, and the gold material of the 585th standard gets a dark brown shade.
Lack of reaction is a sure sign of a fake.
The procedure for checking the authenticity of gold with iodine. Step by step instructions. Video and photo
Description of the method, how to check the gold at home for authenticity with iodine.
Testing is easy and safe. The whole procedure takes place in several stages.





Treat the item with 585 or 583 proofs with medical alcohol
At the first stage of preparation, the product is treated with medical alcohol. After treatment, let it dry.

Sand the metal on the inside
The second preparatory stage is carried out with sandpaper. The jewelry or simply a gold object is sanded on the side that is not "front". So ring is treated on the inside. It is necessary to remove the top layer of metal, as it may be covered by a film of external substances. It is necessary to open the gold layer.
In addition to the sandpaper, you can use any abrasive material, just not very rough, so as not to remove too thick a layer of metal and not to damage the product.
Apply iodine to the jewelry in the rubbed area
The next step is to apply iodine with a cotton swab. You can use a dropper, which can be used to drip exactly on the spot that has been cleaned. You should try to minimize the area on which the iodine hits. There will be a reaction on it and a change in color will occur.
The reaction does not depend on the size of the stain, so you should not try to get more reagent on the surface.

We get the result
Further steps are characterized by observation of the process. A chemical reaction occurs with a substance from the group of halogens, which includes iodine. You can check gold with iodine by the color change. Variations in brown or dark green hues indicate not only that gold is involved in the reaction, the iodine indicates the grade.
Greenish tones characterize a lower grade metal with high copper content.
Table: The results of checking the authenticity of gold with iodine at home
To reliably determine which metals and alloys are involved in chemical reactions, there is a table. It allows you to double-check the type of metallic material by the color of the drop.
A drop or residue of iodine on gold that has reacted (or not) is wiped dry with a soft napkin. Observe, determine the color of the trace left by it.
| Metal or alloy | Droplet Color | Residual stain color |
|---|---|---|
| 385 gold | The drop is dark, gradually turning green | Dark with a dark green tint |
| 585 gold | The drop doesn't turn light, it turns slightly green | Dark |
| Brass | The composition lightens to a white hue | Bright |
| Copper | The composition is getting progressively lighter | Bright |
The method is simple and convenient, it does not require complex reagents and equipment. You can do everything in the kitchen without fear for safety or any chemicals on the objects.
The main disadvantage is that it is impossible to examine the jewelry from the inside, only the surface layer material is determined.
Other means of verification
Among other means to verify the authenticity of precious metal is also a few simple options.
Magnet
Checking with a magnet. The property of gold to be a diamagnetic is used. This is a substance that does not react to a magnetic field. Moreover, the pure metal will repel a magnet from itself. Other metals, with the exception of precious metals, have some degree of ferromagnetic properties, experiencing a mutual attraction with the magnet.
Therefore, low-grade gold with additives of other metals can react to a magnet. If the item "sticks" to it, it is a sign that a ferromagnetic alloy with iron, nickel, or other metals was used.
In this way, you can identify fake jewelry or detect a lower grade at home.
There is a possibility of counterfeit materials such as copper and aluminum, as they are not magnetic. However, in the presence of a strong magnet, there is a field between it and the copper, which is triggered in such a way that when approaching the metal and almost touching it, the magnet stops abruptly.

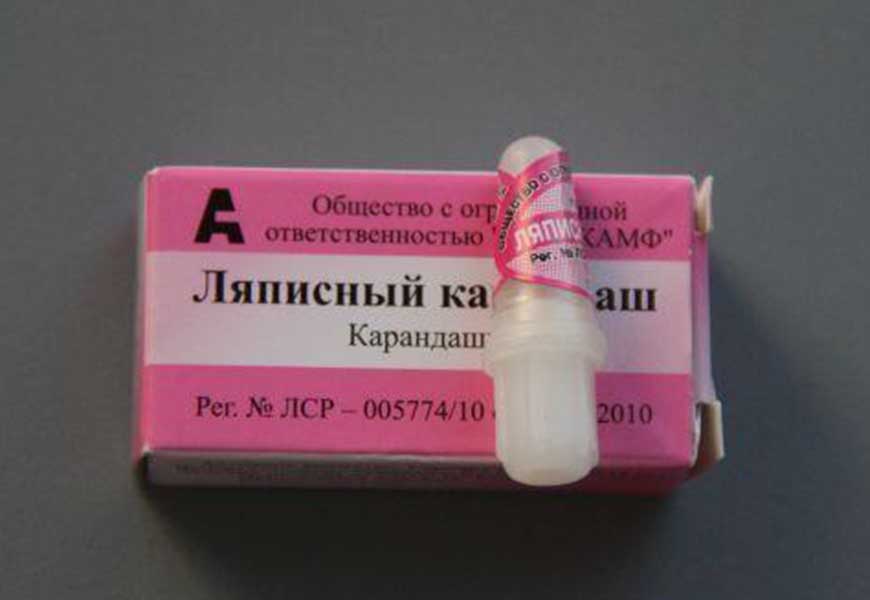
With a lapis pencil
Drug based on nitrates silver and potassium come in handy for testing gold. To do this, the jewelry is dipped under water or wet part of it, and a pencil stripe is applied to the wet pad. Then wipe it with a dry cloth or cotton pad. The trace left by the lapis indicates that the item is counterfeit.
There won't be any streaks left on the gold.
Homemade ways to clean products from iodine
Once you have finished checking the precious metal using iodine solution, you can product cleaning from its residue. Although gold iodide is believed to recover to gold over time, no trace of it should remain. But to clean your jewelry faster, you can resort to one of several methods, using solutions and mixtures.
Hyposulfite: 1 tablespoon of fixer per glass of warm water
Sodium hyposulfite is used in medicine as an antidote and remedy for poisoning, sold in the pharmacy. It is used for the purpose of washing iodine in the form of an aqueous solution - 1 tablespoon per glass of water. Jewelry is in the solution for 30 minutes, rinsed with water, wiped with a soft napkin.

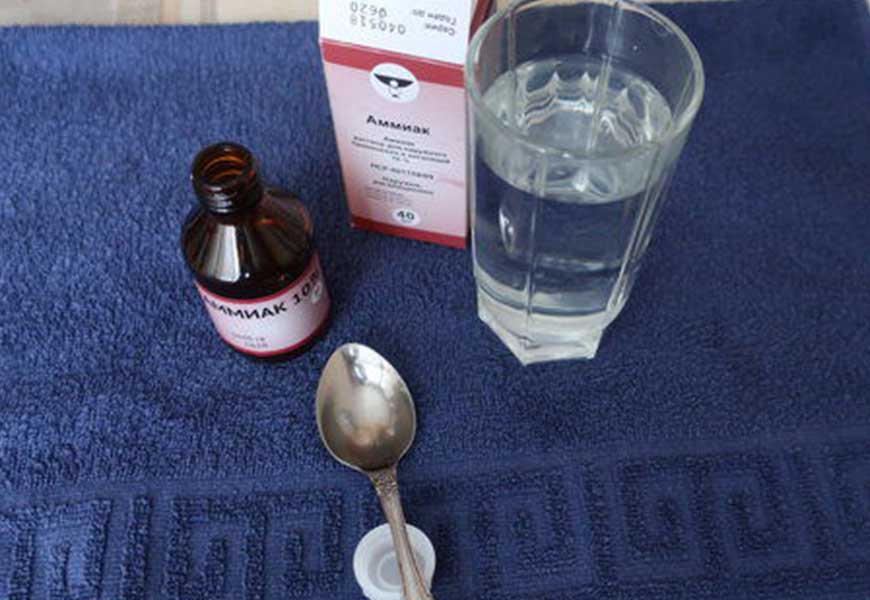
Ammonia solution
Ammonia solution is not effective in all cases, but sometimes it helps. You dip the jewelry in it, and you can add a soapy mixture. Then rinse under running water.
Caution should be exercised if there are stones, although some of them, for example diamonds, only get brighter and acquire additional luster.
A solution of 80 grams of lime with 70 grams of soda and 20 grams of salt in 1 liter of warm water
Another solution to remove traces of iodine: food bicarbonate of soda is supplemented with bleach (bleach) and sodium chloride (table salt). Proportions in grams: 70:80:20. This mixture is dissolved in 1 liter of water and heated. Gold is dipped into the solution.
Watching him, you can see how the iodine comes off the product.
After the procedure, rinse with water, wipe with a damp cloth.

Vinegar
Another household remedy for getting rid of iodine residue. It is better to use table vinegar, not essence, so that you don't get into trouble when it gets on your skin. A cotton swab moistened with the solution is used to wipe the stain all the way dissolution. Rinsed with water and dried.

Coca-Cola
An almost universal cleaner of dirt, as it turns out, is Coca-Cola. You can immerse the entire piece of jewelry, it will remove many varieties of plaque and stains.
If the various means Cleanup did not help, you can simply wait for the iodine streak to go away on its own. As a last resort, you will have to go to a professional jeweler who will solve the problem.

The convenience of the method and its disadvantages
Video instruction: how to test gold jewelry with iodine at home.
Jeweler's services as a reliable and safe way to verify the authenticity of metal
Folk methods of metal testing are good as an express method. For reliability and confidence in the results, it is worth turning to a professional jeweler. In his arsenal will find all the necessary means to determine the authenticity of gold jewelry.
Reviews
We tried a home iodine test on a gold ring that we bought at home. The assay did not inspire confidence, it was a little fuzzy. The method showed that it was real, at least on the surface side. The dark stain did not get any lighter. We did not wash it off, the iodine wore off in a few days.


From time to time I conduct such chemical experiments with gold items. A couple of times I came across strange alloys that were weakly magnetic. Iodine was also incomprehensible, the stain was lighter, but it did not turn white.
I found out a long time ago, tried the method with iodine and more than once. Gives a good result as a quick method, but I still prefer to go to a jeweler. It is more reliable.

